Faculty with primary appointments in the Biomedical Sciences Department.
Assistant Professor, Department of Biomedical Sciences
Phone: 330.325.6682
Email: mansari1@neomed.edu
Assistant Professor, Department of Biomedical Sciences
Email: mazzam1@neomed.edu
Phone: 330-325-6610
Dean, College of Medicine
Professor of Surgery
Professor, Department of Biomedical Sciences
Email: mbasson@neomed.edu
Phone: 330-325-6753
Dr. Basson’s laboratory interests focus on the development of FAK-activating small molecules toward drugs to promote mucosal repair and on the role of SLFN12 in cancer biology. This work includes both cell culture studies and mouse and rat models. He also has extensive clinical research interests in surgical disease and the effects of blood pressure variability on health outcomes.
Professor, Department of Biomedical Sciences
Phone: 330.325.6537
Email: ychen1@neomed.edu
Publications
Our research focuses on the molecular mechanisms underlying redox signal pathway in mediating the disease pathogenesis of myocardial infarction. We are interested in the role of mitochondria-derived oxygen free radicals, signals of glutathione and nitric oxide in regulating oxidative post-translational modifications (OPTM) and how these events impact the bioenergetics function of mitochondria in the post-ischemic heart. One line in our work has established alterations of protein S-glutathionylation in mitochondrial complex I and complex II are linked to mitochondrial dysfunction caused by oxidant stress after myocardial ischemia and reperfusion. In collaboration with the principle investigators of NEOMED/KSU, we are extending the research of this marker to the animal models of type II diabetics and obesity.
A second area of work in the lab is focused on the redox pathway of GSH in controlling the status of complex I/complex II S-glutathionylation and superoxide generation mediated by electron transport chain during myocardial ischemia and reperfusion. The mechanism underlying if protein S-glutathionylation as an indicative of overproduction of superoxide in vivo or not is extensively explored. We employ the pharmacologic approach and genetic modified mouse model to study the mechanism of altering redox modification of mitochondrial proteins. We employed the technique of electron paramagnetic resonance (EPR) to measure the oxygen free radical(s) and redox status of mitochondria in heart.
A third area of work in the lab is focus on how eNOS signal pathway regulates mitochondrial function in the heart and the OPTM of mitochondrial electron transport chain in the post-ischemic heart. In collaboration with the investigators from the OSU, we has established that increasing protein tyrosine nitration of complex I and complex II was detected in the post-ischemic heart, and closely related to overproduction of NO by eNOS in the early phase of reperfusion. Physiologically the signal of NO generated by eNOS can regulate mitochondrial respiration, and further modulates the redox status of mitochondria in myocytes. We employ the genetic modified mouse and generate a new mouse model to investigate how the signal by eNOS regulates complex I/complex II S-glutathionylation, and related mitochondrial dysfunction resulted from post-ischemic injury.
Professor, Department of Biomedical Sciences
Phone: 330.325.6426
Email: wchilian@neomed.edu
Publications
My interest in the vascular biology has been developing for many years along the lines of acute and chronic adaptations of the coronary circulation to physiological and pathophysiological stresses. With regard to chronic adaptations of the coronary circulation, my laboratory was the first to show that ischemia, rather than shear stress, initiates coronary collateral growth. Shear stress is not precluded from this process; rather this mechanism plays a role in the expansion of the vessel after the ischemic stimulus wanes. More recently my laboratory has studied the role the mitochondrial oxidative stress in coronary collateral development, and we have found that such stress blunts the adaptive growth of coronary collaterals. Moreover, rectification of oxidative stress will restore collateral growth in a preclinical model with a poor growth phenotype. With regard to acute adaptations of the coronary circulation to physiological and pathophysiological stresses, I am proud of the accomplishments of my laboratory: we were the first to document myogenic and flow-dependent control of coronary tone; the distribution of microvascular resistance in the beating heart, the effects of preconditioning extended to the vascular endothelium, the mechanism of alpha-adrenergic coronary constriction involves release of a cardiac myocyte derived vasoconstrictor, coronary metabolic dilation is a feed-forward process involving mitochondrial production of H2O2, metabolic dilation is mediated by redox reactions, and Kv1.5 channels play a critical role in metabolic dilation in the heart.
Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6310
Email: lcooper@neomed.edu
Select Publications
The Cooper Lab investigates age-related changes in the skeletons of long-lived bats. Bats as a group are unique in that their bones bend with wingbeats and they display superb resistance to fracture. The Cooper Lab is currently investigating how bats maintain and renew this flexible matrix for bioengineering applications. In addition, the Cooper Lab’s molecular, biomechanical and structural results shows that bats are unusual in that their wing bones lack age-related bone fragility. The Cooper Lab is currently investigating the molecular mechanisms driving the prevention of age-related bone fragility in bats. Genes of bats that were identified as critical to the maintenance of bone integrity in bats replaced those of mice in cell culture studies. The Cooper Lab aims to modify the cells of elderly mice such that they produce a more youthful bone matrix that lacks vulnerabilities that typically lead to fragility diseases.
Associate Professor, Department of Biomedical Sciences
Email: dcoovert@neomed.edu
Phone Number: 330.325.6476
Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6138
Email: fdong@neomed.edu
Publications
My research has focused on exploring the molecular mechanisms underlying cardiovascular diseases, diabetes, and gene and cell therapy for cardiac dysfunction. Additionally, I am responsible for overseeing the Single Cell Genomics facility. Our team examined the role of leptin and adiponectin in obesity-related myocardial dysfunction and established a link between leptin levels and cardiac function. We developed a system with which we can specifically knock down the expression of a gene of interest in White blood cells (WBC)/Hematopoietic stem cells (HSCs). With this system, we found that down-regulation of WBC HIF-1α leads to decreased WBC recruitment and improved left ventricular remodeling following myocardial infarction. In addition, our research reveals a specific paracrine mechanism of Mesenchymal stem cells (MSCs) engraftment. We highlighted the significance of the SDF-1: CXCR4 axis in tissue repair and proposed its exploitation to enhance outcomes in heart failure patients. By developing clinically relevant strategies to manipulate this axis, we can improve outcomes in clinical populations. Moreover, we discovered a new mouse model for aortic valve stenosis, linking CXCR4 deletion in endothelial cells to the development of stenosis and left ventricular hypertrophy. These findings have implications for understanding and potentially treating these cardiac conditions.

Jessica Ferrell, Ph.D.
Assistant Professor, Department of Biomedical Sciences
Phone: 330-325-6468
Email: jfrancl@neomed.edu
www.ferrell-lab.com
The Ferrell Lab investigates the interactions between the liver, intestine, and brain in the development of alcohol-associated liver disease and metabolic syndrome. These organ systems are influenced by the consumption of high fat diets or alcohol, and by circadian disruption like shift work or jet lag, which can worsen disease conditions. Our research goals are to study 1) how alcohol-associated liver disease and metabolic syndrome affect liver function, and 2) how diet and/or circadian disruption contribute to the development of liver disease.
Current work
Project 1
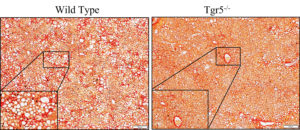
It’s estimated that about 25% of the global population are living with steatosis (fat deposition in the liver), which often occurs along with obesity, Type 2 diabetes, and dyslipidemia. Steatosis, while relatively benign, can progress to steatohepatitis (fatty liver + inflammation), or fibrosis & cirrhosis (scarring) if left untreated. We use special diets to study the transition of steatosis to more severe liver injury states; our goals are to prevent this progression and discover new ways to treat these diseases.
Project 2
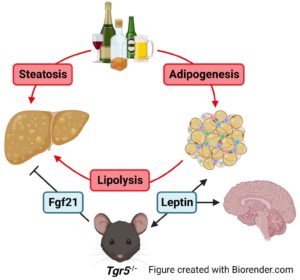
Alcohol consumption can cause steatosis and lead to more severe liver disease like cirrhosis, fibrosis, or alcohol-associated hepatitis, and can also affect the intestine and brain. We are studying the effects of alcohol on liver function as well as the factors that affect alcohol consumption in mice.
Project 3
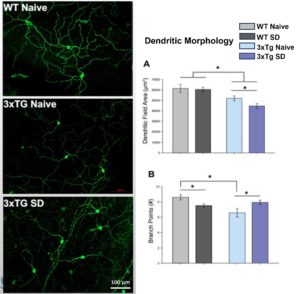
Circadian rhythms direct the timing of our bodies. Disruptions to circadian rhythms (in the form of shift work, jet lag, or sleep loss) can negatively affect organ function and lead to worsened disease states. Currently, we are investigating the effects of long-term sleep deprivation on the progression of conditions like Alzheimer’s disease and metabolic syndrome in mice.
Select publications:
- Pokhrel S, Dilts M, Stahl Z, Boehme S, Frame G, Chiang JYL, Ferrell JM. Tgr5−/− mice are protected from ethanol-induced metabolic alterations through enhanced leptin and Fgf21 signaling. 2023. Commun. 7: e0138. PMID: 37185802.
- Ferrell JM, Dilts M, Stahl Z, Boehme S, Pokhrel S, Wang X, Chiang JYL. Altered serotonin metabolism in Takeda G protein-coupled receptor 5 knockout mice protects against diet-induced hepatic fibrosis. 2022. Res. 6: 214-226.
- Ferrell JM, Chiang JYL. Bile acid receptors and signaling crosstalk in the liver, gut, and brain. 2021. Res. 5: 105-118.
- Chiang JYL, Ferrell JM. Bile acid receptors FXR and TGR5 signaling in fatty liver diseases and therapy. 2020 J. Physiol. Gastrointest. Liver Physiol. 318(3):G554-G573. PMID: 31984784.
- Ferrell JM, Pathak P, Boehme S, Gilliland T, Chiang JYL. Deficiency of both farnesoid X receptor and Takeda G protein-coupled receptor 5 exacerbated liver fibrosis in mice. 2019 Hepatology. 70: 955-970. PMID: 30664797.
- Ferrell JM, Chiang JYL. Understanding bile acid signaling in diabetes: from pathophysiology to therapeutic targets. 2019 Diabetes Metab. J.43(3): 257-272. PMID: 31210034.
- Ferrell JM, Boehme S, Li F, Chiang JY. Cholesterol 7α-hydroxylase-deficient mice are protected from high-fat/high-cholesterol diet-induced metabolic disorders. 2016 Lipid. Res.57: 1144-1154. PMID: 27146480.
- Ferrell JM, Chiang JY. Short-term circadian disruption impairs bile acid and lipid homeostasis in mice. 2015 Cell Mol. Gastroenterol. Hepatol. 1(6): 664-677. PMID: 26645046.
Assistant Professor, Department of Biomedical Sciences
Program Director, Foundations of Medicine Graduate Program
Phone: 330.325.6566
Email: efranks@neomed.edu
Professor, Department of Biomedical Sciences
Phone: 330.325.6640
Email: agalaz@neomed.edu
Select Publications
The major focus of our research is tinnitus, the sensation of hearing a sound when no external sound is present. Almost all individuals experience this sensation for brief, unobtrusive periods. However, chronic sensation of tinnitus affects approximately 17% of the general US population. People with severe tinnitus may have trouble hearing, working and sleeping. Despite its ubiquity, the pathophysiology of tinnitus is poorly understood, and there is no FDA approved cure or treatment. Using the mouse tinnitus model which we have developed in our laboratory we study brain mechanisms responsible for tinnitus induction following an acoustic trauma. For our research we use a wide variety of techniques spanning from recording of electrical activity in single neurons extracellularly as well as intracellularly to behavioral approaches of tinnitus assessment using gap-induced inhibition of the acoustic startle reflex.
We thank the National Institute for Deafness and other Communication Disorders for their support of this work.
Professor, Department of Biomedical Sciences
Phone: 330.325.6299
Email: rgerman@neomed.edu
Select Publications
Swallowing difficulties, and the failure to protect the airway, are a major cause of health problems in premature or preterm infants. The neurological cause for these problems is unknown, and as a result there are few effective therapies. In our lab we work on understanding the biomechanics and pathology of neural control of swallowing using an animal model, the baby pig. Understanding the mechanism of airway protection failure will provide a biological basis for decisions about care and intervention in these fragile and cherished patients.
Another focus in the lab is the impact of Parkinson’s disease on feeding, swallowing, and airway protection. Such patients suffer from many effects of compromised eating, such as reduced nutrition and chronic aspiration. These problems hard to diagnose because patients do not report them and they require invasive imaging to be seen. In collaboration with Dr. Jason Richardson, we are carrying out animal model, integrative studies from brain to tongue and jaws, to determine what is going wrong, and how it can be fixed.
Adam G. Goodwill, Ph.D., F.C.V.S.
Assistant Professor, Department of Biomedical Sciences
Phone: 330.325.6886
Email: agoodwill@neomed.edu
Research in our group focuses on the interplay between myocardial contractile function and delivery of oxygen/nutrients via coronary blood flow. In other words, we study how the heart balances delivery of fuel (blood) with its current demands. Our overall goal is to improve the understanding of key regulators that link oxygen demand with delivery while also exploring how this delicate balance can become disrupted in disease states. Our laboratory employs a top-down approach, beginning at the level of dysfunction and investigating which dysregulated mediators may be responsible for the imbalances associated with cardiac pathologies. Our studies routinely begin in vivo and progress to the molecular. Current work is focused on identifying the mechanisms responsible for cardioprotective phenomena found with certain drug classes. The goal of this work is to identify therapeutic targets for the treatment of cardiovascular disease.
Publications
- Baker HE, Tune JD, Mather KJ, Blaettner BS, Clark HE, Li F, Li X, Kowala MC, Fliegel L, Goodwill AG. Acute SGLT-2i treatment improves cardiac efficiency during myocardial ischemia independent of Na(+)/H(+) exchanger-1. Int J Cardiol. 2022 Sep 15;363:138-148. doi: 10.1016/j.ijcard.2022.06.054. Epub 2022 Jun 23. PubMed PMID: 35753619.
- Goodwill AG, Baker HE, Dick GM, McCallinhart PE, Bailey CA, Brown SM, Man JJ, Tharp DL, Clark HE, Blaettner BS, Jaffe IZ, Bowles DK, Trask AJ, Tune JD, Bender SB. Mineralocorticoid receptor blockade normalizes coronary resistance in obese swine independent of functional alterations in K(v) channels. Basic Res Cardiol. 2021 May 20;116(1):35. doi: 10.1007/s00395-021-00879-3. PubMed PMID: 34018061; PubMed Central PMCID: PMC8552965.
- Tune JD, Baker HE, Berwick Z, Moberly SP, Casalini ED, Noblet JN, Zhen E, Kowala MC, Christe ME, Goodwill AG. Distinct hemodynamic responses to (pyr)apelin-13 in large animal models. Am J Physiol Heart Circ Physiol. 2020 Apr 1;318(4):H747-H755. doi: 10.1152/ajpheart.00365.2019. Epub 2020 Feb 28. PubMed PMID: 32108522.
- Baker HE, Kiel AM, Luebbe ST, Simon BR, Earl CC, Regmi A, Roell WC, Mather KJ, Tune JD, Goodwill AG. Inhibition of sodium-glucose cotransporter-2 preserves cardiac function during regional myocardial ischemia independent of alterations in myocardial substrate utilization. Basic Res Cardiol. 2019 Apr 19;114(3):25. doi: 10.1007/s00395-019-0733-2. PubMed PMID: 31004234; PubMed Central PMCID: PMC6616532.
- Sassoon DJ, Tune JD, Mather KJ, Noblet JN, Eagleson MA, Conteh AM, Sturek JT, Goodwill AG. Glucagon-Like Peptide 1 Receptor Activation Augments Cardiac Output and Improves Cardiac Efficiency in Obese Swine After Myocardial Infarction. Diabetes. 2017 Aug;66(8):2230-2240. doi: 10.2337/db16-1206. Epub 2017 May 8. PubMed PMID: 28483802; PubMed Central PMCID: PMC5521862.
- Goodwill AG, Dick GM, Kiel AM, Tune JD. Regulation of Coronary Blood Flow. Compr Physiol. 2017 Mar 16;7(2):321-382. doi: 10.1002/cphy.c160016. PubMed PMID: 28333376; PubMed Central PMCID: PMC5966026.
- Goodwill AG, Noblet JN, Sassoon D, Fu L, Kassab GS, Schepers L, Herring BP, Rottgen TS, Tune JD, Dick GM. Critical contribution of KV1 channels to the regulation of coronary blood flow. Basic Res Cardiol. 2016 Sep;111(5):56. doi: 10.1007/s00395-016-0575-0. Epub 2016 Aug 5. PubMed PMID: 27496159; PubMed Central PMCID: PMC5193223.
- Goodwill AG, Tune JD, Noblet JN, Conteh AM, Sassoon D, Casalini ED, Mather KJ. Glucagon-like peptide-1 (7-36) but not (9-36) augments cardiac output during myocardial ischemia via a Frank-Starling mechanism. Basic Res Cardiol. 2014;109(5):426. doi: 10.1007/s00395-014-0426-9. Epub 2014 Jul 9. PubMed PMID: 25005062; PubMed Central PMCID: PMC4259250.
Assistant Professor of Biomedical Sciences
Office: D-104
Email: ngriderpotter@neomed.edu
RESEARCH
My research focuses on comparative primate locomotion and musculoskeletal functional morphology.
Professor, Department of Biomedical Sciences
Phone: 330.325.6704
Email:
Select Publications
Dr. Haqqi’s group focuses on developing new treatment modalities for degenerative joint diseases such as Osteoarthritis. Aging is a major factor for chronic diseases including osteoarthritis, a leading cause of joint dysfunction associated with cartilage
degradation, disability and poor quality of life in the affected population worldwide. Among adults 60 years of age or older the prevalence of symptomatic knee osteoarthritis is approximately 10 percent in men and 13 percent in women. There are no disease-modifying medical therapies currently available for osteoarthritis. The ultimate objective of our research program is to address this unmet need, by identifying and validating novel compounds and their target molecules in chondrocytes, the only cell type present in the cartilage, that can inhibit induction and/or limit the progression of osteoarthritis. In addition, the group is also studying epigenetics in cartilage and the potential of plant derived inhibitors that will be most effective in suppressing joint damage. These research projects are funded by the NIH/National Institutes of Arthritis and Musculoskeletal Diseases and the NIH/National Center for Complementary and Integrative Health.
Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6635
Email: thieronymus@neomed.edu
Select Publications
Evolutionary Morphology of Bird Wings
My research aim is to understand the musculoskeletal anatomy and function of bird wings. Birds have dramatically altered the common components of the forelimb to respond to the functional demands of flight. Within this system, my research addresses three basic questions: (1) what are the morphological adaptations that allow birds to fly so efficiently (or, what can we learn from birds about building wings)? (2) how have birds modified basic tissues such as bone and ligament to adapt to new forms of loading (what can bird bones teach us about material design)? (3) how has the evolution of different avian flight styles and capabilities played out over evolutionary time (some wing morphology is adaptive for specific types of flight, some may be phylogenetic inertia—which parts are which)? My lab addresses these questions using novel techniques that bridge the gap in scale between standard gross anatomy and histology, as well as modeling and analysis approaches that allow us to leverage the diversity of living and fossil birds as a broad pool of natural experiments.
Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6415
Email: ylee3@neomed.edu
My research interests are the role of orphan nuclear receptor SHP in diet induced diabetes and obesity.
Assistant Professor of Biomedical Sciences
Office: RGE-334
Associate Professor of Biomedical Sciences
Office: RGE-140
Phone: 330-325-6810
Email: gliu@neomed.edu
Research interests
- Investigate the pathogenesis of lower urinary tract dysfunction using animal models.
- Examine the mechanisms of urological complications in obesity and diabetes, including bladder dysfunction, urinary incontinence, erectile dysfunction, and urinary tract infection
- Develop new treatments for gastrointestinal injury
Professor of Neurobiology, Department of Biomedical Sciences
Phone: 330.325.6656
Email: ylu@neomed.edu
Select Publications
We investigate the cellular mechanisms of auditory processing, with a focus on neuromodulation under normal hearing and hearing-impaired conditions. Electrophysiological (e.g. whole-cell recordings) and optical imaging (e.g. calcium imaging) approaches, combined with pharmacological immunohistochemistry tools, are employed. We aim to provide a basic understanding of neuromodulation in functionally well-established auditory circuits that analyze information for sound localization. Ultimately, this will provide the basis for therapeutic intervention in hearing disorders characterized by impaired sensitivity to precise temporal features in sounds.
Assistant Professor, Department of Biomedical Sciences
Phone: 330.325.5916
Email: jmellott@neomed.edu
Select Publications
We study how the neural circuits in hearing change as we age. Age-related hearing loss is associated with a reduction in the level of GABA, a key neurochemical used to communicate among neurons throughout the auditory system. The loss of GABA leads to a variety of hearing deficits, including impairment of the ability to detect fine differences in the timing of sounds.
My laboratory identifies the auditory circuits particularly susceptible to GABA loss during aging, using complex circuit-tracing, immunohistological and imaging methods.

Vahagn Ohanyan, M.D., Ph.D.
Associate Professor, Department of Biomedical Sciences
RGE-338 / 330-325-6535
vohanyan@neomed.edu
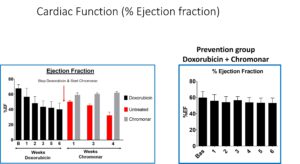
In our laboratory, we are dedicated to investigating the intricate and often poorly understood phenomenon of coronary microvascular dysfunction that occurs in response to chemotherapy. Our primary focus centers on unraveling the mechanisms underlying doxorubicin-induced cardiotoxicity while simultaneously exploring novel treatments and preventive measures to safeguard the heart.
To accomplish these objectives, we have developed a robust preclinical mouse model that mimics the development of heart failure induced by chemotherapy. This model allows us to closely study the pathophysiological changes that occur in the coronary microvasculature and myocardium during chemotherapy administration.
A significant facet of our research involves the utilization of Chromonar, an established medication recognized for its coronary-specific vasodilatory properties. By harnessing the vasodilatory effects of Chromonar, we aim to enhance myocardial blood flow in the presence of doxorubicin, thereby preventing or mitigating the cardiotoxic effects associated with this widely used chemotherapy agent.
Our research endeavors are multifaceted, encompassing detailed molecular and cellular investigations, as well as comprehensive physiological assessments. Through meticulous experimentation and data analysis, we are working to unravel the intricate interplay between chemotherapy, coronary microvascular dysfunction, and the potential protective mechanisms conferred by Chromonar.
The ultimate goal of our research is to not only enhance our understanding of the complex relationship between chemotherapy and cardiac health but also to contribute to the development of more effective therapeutic strategies and prevention methods for chemotherapy-induced cardiotoxicity. By shedding light on these critical issues, we hope to improve the overall quality of life for cancer patients undergoing chemotherapy while ensuring the long-term well-being of their cardiovascular health.
Recent publications:
- Anna Winnicki, James Gadd, Vahagn Ohanyan,Gilbert Hernandez,Yang Wang,Molly Enrick,Hannah McKillen,Matthew Kiedrowski, Dipan Kundu,Karlina Kegecik,Marc Penn,William Chilian,Liya Yin*,Feng Dong* Role of Endothelial CXCR4 in the Development of Aortic Valve Stenosis Original Research, Front. Cardiovasc. Med. – Cardiovascular Therapeutics, DOI: 10.3389/fcvm.2022.971321 Manuscript Id: 971321
- Feng Dong M.D., Ph.D1.,* Liya Yin M.D., Ph.D. 1, Hamayak Sisakian MD., PhD., 2 Tatevik Hakobyan MD., 1 Lacey S. Jeong M.D.1, Hirva Joshi M.D1., Ellianna Hoff M.D1., Selena Chandler M.D1., Geetika Srivastava M.D1., Abdur Rahman Jabir B.S1., Kelly Kimball B.S. 1, Yeong-Renn Chen Ph.D. 1, Chwen-Lih Chen BS1, Patrick T. Kang Ph.D. 1, Parisa Shabani Ph.D. 1, Lindsay Shockling B.S. 1, Thomas Pucci M.S. 1, Karlina Kegecik B.S. 1, Christopher Kolz1, William M. Chilian Ph.D1. and Vahagn Ohanyan M.D., Ph.D1* Takotsubo Syndrome is A Disease of the Coronary Microcirculation- ( European Heart Journal) – (Drs Feng Dong and Vahagn Ohanyan are Co-Corresponding Authors)
- Enrick M, Jamaiyar A, Ohanyan V, Juguilon C, Kolz C, Shi X, Janota D, Wan W, Richardson D, Stevanov K, Hakobyan T, Shockling L, Diaz A, Usip S, Dong F, Zhang P, Chilian WM, Yin L The Roles of Bone Marrow-Derived Stem Cells in Coronary Collateral Growth Induced by Repetitive Ischemia. Cells. 2023 Jan 6;12(2):242. doi: 10.3390/cells12020242.PMID: 3667217
- Khanal S, Bhavnani N, Mathias A, Lallo J, Gupta S, Ohanyan V, Ferrell JM, Raman P. Deletion of Smooth Muscle O-GlcNAc Transferase Prevents Development of Atherosclerosis in Western Diet-Fed Hyperglycemic ApoE-/- Mice In Vivo.Int J Mol Sci. 2023 Apr 26;24(9):7899. doi: 10.3390/ijms24097899.PMID: 37175604
Ravi K. Adapala1#, Venkatesh Katari1#, Anantha K. Kanugula2, Vahagn Ohanyan2, Sailaja Paruchuri1 and Charles K. Thodeti1* Deletion of endothelial TRPV4 protects heart from pressure-overload-induced hypertrophy. Hyperte
Associate Professor of Physiology and Pharmacology, Department of Biomedical Sciences
Phone: 330.325.6425
Email: praman@neomed.edu
Elucidation of cellular and molecular mechanism(s) of accelerated atherogenesis among diabetics is the primary focus of my research.
In the light of the importance of obesity as an independent risk-factor for both diabetes and related vascular complications, my research program is two-fold: 1) regulation of vascular gene expression by adipokines to better comprehend the role of the adipose tissue in vascular diabetic complications, and 2) to explore the underlying MOA of antidiabetic agents implicated in diabetes and related vascular disorders.
Currently, we are studying the role of thrombospondin-1 (TSP-1), a potent antiangiogenic and proatherogenic protein with strong implications in atherogenesis, in leptin-mediated macrovascular complications.
We are using various approaches that involve cell and molecular biology, biochemistry and mouse models for these studies.
Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6516
Email: mrosen@neomed.edu
Select Publications
Early hearing loss is a risk factor for later problems with speech processing due to changes in auditory brain regions, and early stress exacerbates these deficits. Our laboratory studies how early hearing loss and stress can change auditory perception related to speech, and its underlying neural circuits. We use behavioral, neurophysiological, neurochemical, anatomical, and computational techniques to measure how neural activity and circuitry are altered by these developmental disorders, and how neural changes correlate with deficits in learning and perception. We measure neural responses to vocal communication and to natural and artificial sounds in the Mongolian gerbil, using implanted electrode arrays or intracellular recordings, where we can manipulate neurochemicals to assess circuit contributions to neural responses. We test animal perception using a variety of behavioral tasks, including operant conditioning and acoustic startle. This work lets us identify interventions that remediate perceptual deficits arising from these early experiences, in addition to understanding their underlying causes.
Professor, Department of Biomedical Sciences
Ohio Research Scholar
Director of Musculoskeletal Research Group
Phone: 330.325.6619
Email: fsafadi@neomed.edu
Select Publications
Regulation of bone cell development and function, with specific emphasis on growth factors that can enhance osteogenesis
Bone loss is a major health care problem in the United States and worldwide. Risk factors associated with osteoporosis, include estrogen-deficiency (post-menopuasal) and aging. During bone development and its maintenance, the antagonistic processes of bone formation and resorption are regulated by various systemic and local factors (hormones, growth factors, cytokines, etc). It is evident that bone formation during skeletal modeling/remodeling and fracture repair requires stringent control of osteoblast proliferation and differentiation. Regulation of these biological processes involves sequential expression of cell growth and tissue specific genes in response to different regulatory signals.
Dr. Safadi’s research laboratory focuses on the regulation of bone cell development and function, with specific emphasis on growth factors that can enhance osteogenesis. We are interested in various metabolic bone diseases such as age/estrogen-induced osteopenia, osteopetrosis and cartilage-associated diseases (osteoarthritis and rheumatoid arthritis). The goal of our research is understand the pathological mechanisms underlying various bone diseases in order to develop strategies for the therapeutic management of such diseases. We identified a novel growth factor (named Osteoactivin) that has anabolic effects on bone. If we understand the mechanisms responsible for this effect, then this factor could be used as potential therapeutic agent to stimulate bone formation in diseases associated with osteopenia, increased risk of fracture and spinal fusion.
In addition, we developed an interest on the role of osteoactivin (gpnmb) as a neuroprotective factor for neurodegenerative diseases such as Parkinson’s disease and Alzheimer disease. This research is conducted in collaboration with the neurodegenerative research group at NEOMED. We also interested on the role of gpnmb as anti-inflammatory factor in acute and chronic conditions for wound healing and neuroinflammation.
Dr. Safadi’s laboratory is also working on another collaborative research with Dr. Mary Barbe at Temple University School of Medicine. Dr. Barbe laboratory utilizes an in vivo model for cumulative trauma disorder that focuses on the molecular and cellular mechanisms associated with tendonitis and inflammation-induced bone loss.
Professor, Department of Biomedical Sciences
Phone: 330.325.6655
Email: bschofie@neomed.edu
Select Publications
We study brain circuits that modulate how we hear. Such modulation allows us to maximize sensitivity when we need to hear a faint sound (Was that your phone?), filter out noise when we want to hear a friend’s voice in a noisy restaurant, or ignore irrelevant sounds (but not important ones!) when we’re trying to sleep. Acetylcholine is a neurotransmitter that plays a role in these and many other aspects of hearing, helping the brain adapt during normal development, during aging and in response to damage of the ear or brain. A goal of our research is to understand modulation of hearing and how acetylcholine circuits contribute to these tasks.
We study hearing circuits in guinea pigs, rats and mice. We combine the latest techniques using replication-deficient viruses and transgenic animals with classical anatomical tracing and immunohistochemistry to label specific neural pathways. We examine these pathways with light and electron microscopes to identify the cell types and their synaptic connections and thus characterize the brain circuits that allow us to hear.
Associate Professor, Department of Biomedical Sciences
Email: rseese@neomed.edu
330-325-6318
LinkedIn
Research Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6549
Email: sshanbhag@neomed.edu
Select Publications
What are the mechanisms by which information is encoded in the brain and how is this information used? These two questions form the basis of my research. I am currently exploring the representation of social vocalizations by neurons in the amygdala using optogenetic and neurophysiological techniques. I also have a long-standing interest in the coding of spatial information by the auditory system.
Research Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6536
Email: xshi@neomed.edu
Professor, Department of Biomedical Sciences
Email: htawfik@neomed.edu
Phone: 330-325-6642
Ingalls-Brown Professor of Anatomy, Department of Biomedical Sciences
Phone: 330.325.6295
Email: thewisse@neomed.edu
Select Publications
Mammalian Anatomy, Development, and Evolution
Dr. Thewissen’s research centers on the anatomical specializations of whales and combines gross anatomical, histological, embryological, and paleontological methods. The evolution of whales from living on land to living in water as well as the biology of Arctic whales are central stage in the research program. Examples of specific projects include studying whether living whales are deaf by counting nerve cells in their ears, figuring out how old a whale is by studying tree-ring like structures in the skull, and using the fine structure of the teeth to learn about the biology of the animal. Much of the research is based on fossil whales from Pakistan and India, and samples of modern whales from the Alaskan Arctic, both unique resources.
Professor, Department of Biomedical Sciences
Associate Dean of Research, College of Medicine
Phone: 330.325.6630
Email: jjw@neomed.edu
Select Publications
Our laboratory studies neural mechanisms underlying hearing and acoustically guided behaviors, particularly social communication. We have focused increasingly on interactions between hearing and emotions. Much of our current work examines three facets of these interactions: the link between vocal signals and an animal’s emotional state, the analysis of social vocalizations by emotional centers in the brain, and the manner in which emotional centers modify processing of sounds by the auditory system. We use a wide variety of approaches in our work—acoustic, behavioral, neurophysiological, pharmacological, and anatomical. We are particularly excited about technical developments that will allow us to analyze the activity of individual neurons during social interactions. Our work on the basic neural mechanisms forms the basis for future studies of disorders of acoustic communication that involve misinterpretations of the meaning of sounds.
We thank the National Institute for Deafness and other Communication Disorders for their support of this work.
Assistant Professor, Department of Biomedical Sciences
Phone: 330-325-6114
Email: bwinters@neomed.edu
The ability to localize sounds is critical to survival for most animals and in humans facilitates selective attention. The location of sound sources must be computed in the central auditory system from basic frequency, timing, and intensity information. Our brain extracts cues for horizontal (azimuth) sound source location through parallel processing of this information in specialized brainstem nuclei. Because of the strong evolutionary pressure on sound localization ability, the neurons in these nuclei have acquired astounding cellular adaptations that underlie their functional roles within these circuits. Our lab seeks to understand the cellular components of sound localization circuits and how they are sculpted by sound driven activity during development using electrophysiological and imaging approaches. Neurons receive many of their synaptic on tree-like structures called dendrites. However, dendrites do not just passively receive signals. Their physical structure and ion channel composition determines how synaptic signals interact thus determining the input/output transformations of the neuron. We study signal processing in dendrites using dual dendritic patch-clamp combined with multiphoton imaging techniques.
Assistant Professor of Biomedical Sciences
Office: F-238
Phone: 330-325-6418
Email: jwu3@neomed.edu
National Institutes of Health
Google Scholar
Biography:
I completed postdoc training at Virginia Commonwealth University and the University of Connecticut, where I was promoted to Assistant Research Professor. In 2019, I joined the Cleveland Clinic as Project Staff in the Department of Inflammation and Immunity under the mentorship of Dr. Laura Nagy, a distinguished researcher in alcohol research. I was also appointed Assistant Professor in the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University.
I have been investigating liver injury and its underlying mechanisms for over 10 years, focusing on cell metabolism, signaling pathways, and innate immunity in alcohol-associated and other chronic liver diseases. My research has been funded by the NIH and the Cleveland Digestive Diseases Research Core Center. With a broad background in liver pathophysiology and extensive experience in defining gene expression, function, and regulation, I have always been passionate about and dedicated to biomedical research and education.
Associate Professor, Department of Biomedical Sciences
Phone: 330.325.6304
Email: jwyoung@neomed.edu
Select Publications
Research in the Young Lab focuses on evolutionary, comparative and developmental aspects of mammalian locomotion. Our work is question driven and is currently concentrated on two topics: the biomechanics of arboreal locomotion, particularly in primates, and the interaction between musculoskeletal growth and locomotor development.
A principal focus of the work in the lab has been the biomechanics of arboreal locomotion in primates. The aim of this work is to relate standard biomechanical measures – including gait patterns, joint postures, limb forces and center of mass movements – to fitness-critical variables such as balance, accelerative capacity and energetic efficiency. A variety of techniques are used to address this aim, including three-dimensional motion tracking, measurement of single-limb kinetics and whole-body mechanics using custom-designed force transducers, and morphometric assessments of musculoskeletal anatomy. Our current research focuses on using state-of-the-art techniques to quantify locomotor kinematics in free-ranging primates moving in their natural habitats.
A second focus of our research in the lab has been the degree to which growth and development might be adaptively constrained to promote fitness across the lifespan. Immature mammals must often compete against adults for resources, evade common predators and keep pace during group travel, despite small body size, an underdeveloped musculoskeletal system and other limits on performance. We should expect strong selection for mechanisms that permit young individuals to overcome such limitations and reach reproductive maturity. Previous research has examined how allometric changes in skeletal form and locomotor mechanics might facilitate improved locomotor performance in young mammals. Our current research i is focused on how natural selection has impacted growth and locomotor development in cottontail rabbits, a model system representing fast-growing ecologically challenged mammals.
Associate Professor of Molecular Pharmacology, Department of Biomedical Sciences
Phone: 330.325.6412
Email: jyun@neomed.edu
Professor and Watanakunakorn Chair, Department of Biomedical Sciences
Professor of Surgery
Phone: 330.325.6342
Email: pzhang@neomed.edu
Contact
Deborah Heeter
Email | 330.325.6636
Department Chair
J.G.M. ‘Hans’ Thewissen, Ph.D.
Phone: 330.325.6295
Email: thewisse@neomed.edu
Department of Biomedical Sciences
Department of Biomedical Sciences home
College of Medicine home page

